Highly Efficient Perovskite Solar Cells using Sheet-shaped Molecule: Increasing Power Conversion Efficiency by 1.2 Times Compared with Conventional Material
|
Published in “Journal of the American Chemical Society“(Online Publication, December 10, 2015). |
Prof. Murata, Y., Assoc Prof.Wakamiya, A., |
|||
|
Assoc Prof. Wakamiya, A., Mr. Nishimura, H., Ms. Shimazaki, I., Prof. Murata, Y. (Structural Organic Chemistry, Division of Synthetic Chemistry)
Assoc Prof. Saeki, A., Mr. Ishida, N. (Graduate School of Engineering, Osaka University)
Prof. Scott, L, T. (Merkert Chemistry Center, Boston College, USA) |
||||
|
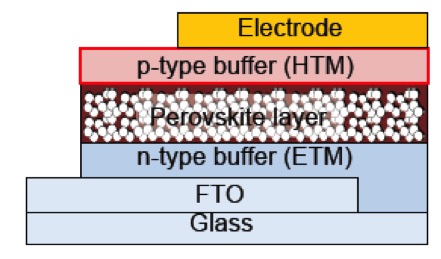
Perovskite solar cells represent attractive cost-effective next generation printable photovoltaics. In this type of solar cells, perovskite semiconductors, such as lead halide perovskite CH3NH3PbI3, act as photoabsorbers to generate free carriers that can be collected in the electrodes through both p- and n-type buffer layers (Figure 1). In a relatively short period, power conversion efficiencies (PCEs) in such cells have been substantially increased, mainly due to improvements of the fabrication protocols for the perovskite layer1). Although tremendous efforts have been devoted to the development of improved hole-transporting materials (HTMs) for buffer layers, the number of high- performance HTMs is still limited. Therefore, 2,2′,7,7′-tetrakis- (N,N-di-p-methoxyphenylamine)-9,9′-spirobifluorene (Spiro-OMeTAD), which is traditionally used for solid-state dye- sensitized solar cells, remains, despite its high cost, the standard buffer material even in perovskite solar cells. For further improvements of the PCE, the development of more cost-effective next-generation HTMs based on new molecular design concepts, and the elucidation of the key factors that determine the photovoltaic performance in perovskite solar cells, will be crucial.
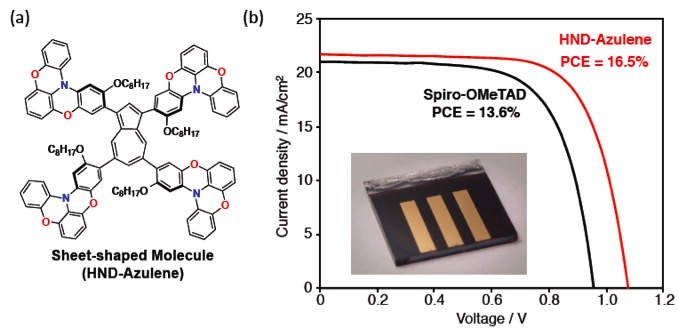
Conventional HTMs for perovskite solar cells are often based on triarylamine skeletons with propeller-like structures. Wakamiya et al. have recently reported that the use of partially oxygen-bridged triarylamines as quasiplanar scaffolds for charge-transporting materials facilitates delocalized π-conjugation and on-top π-stacking in the solid state, which ensures high carrier mobilities in the π-stacking direction.2) In this study, The research group designed and synthesized the two-dimensionally expanded system (HND-Azulene) with a sheet-shaped structure, in which four oxygen-bridged triaryl-amines that contain alkoxy groups to enhance the solubility were attached to a core skeleton (Figure 2a). For the core skeleton, they selected azulene, which is a nonbenzenoid aromatic hydrocarbon that exhibits unique electron-donating and accepting character arising from the five- and seven-membered ring, respectively.
Perovskite solar cells that use HND-Azulene as a HTM exhibited a superior performance (PCE <= 16.5%) relative to those using the current standard Spiro-OMeTAD (Figure 2b). Based on the evaluation and a comparison of the optoelectronic and electrochemical properties of HND-Azulene and Spiro-OMeTAD, they were able to elucidate the factors that are required for HTMs to act efficiently in perovskite solar cells. In line with these new molecular design principles, they believe that several more sophisticated HTMs for perovskite solar cells should be easily attainable. Development and optimization and of more extended HTMs are currently in progress in thier laboratory and results will be reported in due course. |
||||
|
Figure 1.Typical device structure of perovskite solar cells (cross-sectional image) |
||||
|
Figure 2.(a) Sheet-shaped organic semiconductor (HND-Azulene) and (b) the performance of peroskite solar cells using HND-Azulene and the current standard material.
|
||||
|
This study was partially supported by the PRESTO, COI, and ERATO programs from the Japanese Science and Technology Agency (to A. Wakamiya), and by the US National Science Foundation and Department of Energy (to L. T. Scott). The authors would like to thank Prof. Kenichiro Itami (Nagoya University) for fruitful discussions, and Prof. Kazunari Matsuda and Mr. Feijiu. Wang (Kyoto University) for advice on the measurement of SEM images. |
||||
|
References 1) For example, Wakamiya, A.; Endo, M.; Sasamori, T.; Tokitoh, N.; Ogomi, Y.; Hayase, S.; Murata,Y., Reproducible Fabrication of Efficient Perovskite-based Solar Cells: X-ray Crystallographic Studies on the Formation of CH3NH3PbI3Layers, Chem. Lett., 43,711 (2014). |
||||
|
2)Wakamiya, A.; Nishimura, H.; Fukushima, T.; Suzuki, F.; Saeki, A.; Seki, S.; Osaka, I.; Sasamori, T.; Murata, M.; Murata, Y.; Kaji, H. On-Top π-Stacking of Quasiplanar Molecules in Hole-Transporting Materials: Inducing Anisotropic Carrier Mobility in Amorphous Flms, Angew. Chem., Int. Ed., 53,5800 (2014). |
||||
 Institute for Chemical Research, Kyoto University
Institute for Chemical Research, Kyoto University International Joint Usage Research Center
International Joint Usage Research Center