Drs LONG, Youwen ; Assistant Professor SAITO, Takashi ; Associate Professor AZUMA, Masaki ; Professor SHIMAKAWA, Yuichi, “Temperature-induced A-B intersite charge transfer in an A-site-ordered LaCu3Fe4O12 perovskite ” (Published in “Nature “, 5 March 2009)
| Drs LONG, Youwen; Assistant Professor SAITO, Takashi; Associate Professor AZUMA, Masaki; Professor SHIMAKAWA, Yuichi (Laboratory of Advanced Solid State Chemistry, International Reserch Center for Elements Science)
“Temperature-induced A-Bintersite Published in “Nature”, 5 March 2009 |
 Assistant Professor SAITO, Takashi Professor SHIMAKAWA, Yuichi Drs LONG, Youwen Associate Professor AZUMA, Masaki (from left side) |
||
|
Drs. Y.W. Long, T. Saito (Assist. Prof.), M. Azuma (Assoc. Prof.), and Y. Shimakawa (Prof.) in Advanced Solid State Chemistry Laboratory succeeded in preparing a new A-site ordered perovskite-structure compound LaCu3Fe4O12, which shows “temperature-induced intersite charge transfer”. |
|||
|
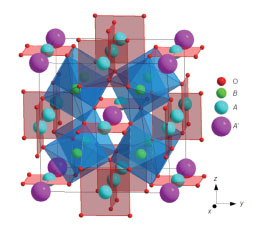
Changes of valence states in transition-metal oxides often cause significant changes in their structural and physical properties. Chemical doping is the conventional way of modulating these valence states. In ABO3perovskite and/or perovskite-like oxides, chemical doping at the A site can introduce holes or electrons at the Bsite, giving rise to exotic physical properties like high-Tc superconductivity and colossal magnetoresistance. When valence-variable transition metals at two different atomic sites are involved simultaneously, we expect to be able to induce charge transfer -and, hence, valence changes- by using a small external stimulus rather than by introducing a doping element. Materials showing this type of charge transfer are very rare, however, and such externally induced valence changes have been observed only under extreme conditions like high pressure. We found unusual temperature-induced valence changes at the A and B sites in the A-site-ordered double perovskite LaCu3Fe4O12; the underlying intersite charge transfer is accompanied by considerable changes in the material’s structural, magnetic and transport properties. When cooled, the compound shows a first-order, reversible transition at 393K from LaCu2+3Fe3.75+4O12with Fe3.75+ ions at the B site to LaCu3+3Fe3+4O12 with rare Cu3+ ions at the A site. Intersite charge transfer between the A-site Cu and B-site Fe ions leads to paramagnetism-to-antiferromagnetism and metal-to-insulator isostructural phase transitions. What is more interesting in relation to technological applications is that this above-room-temperature transition is associated with a large negative thermal expansion. |
|
||
|
The present findings have great impacts on not only research fields of fundamental solid state physic and chemistry but also technological application fields of new materials with new functions. |
|||
|
The research was done in collaboration with N. Hayashi (Assist. Prof.) and S. Muranaka (Prof.) in Graduate School of Human and Environmental Studies, Kyoto University. The results were published in Nature (March 5, 2009) and will also be presented at meetings of the American Physical Society and the Physical Society of Japan. |
|||
 Institute for Chemical Research, Kyoto University
Institute for Chemical Research, Kyoto University International Joint Usage Research Center
International Joint Usage Research Center