First Observation of Face-on Thin Film Structure of Pentacene on Silicon
Published in “Scientific Reports” (Online Publication, January 24, 2019).
(Laboratory of Chemistry for Functionalized Surfaces, Division of Environmental Chemistry)
Assist Prof. Murdey, R., Mr. Nakao, K.
(Laboratory of Molecular Aggregation Analysis, Division of Multidisciplinary Chemistry)
Prof. Yoshida, H.
(Graduate School of Engineering and Molecular Chirality Research Center, Chiba University)
Dr. Koganezawa, T.
(Japan Synchrotron Radiation Research Institute)
Assoc Prof. Eda, K.
(Graduate School of Science, Kobe University)
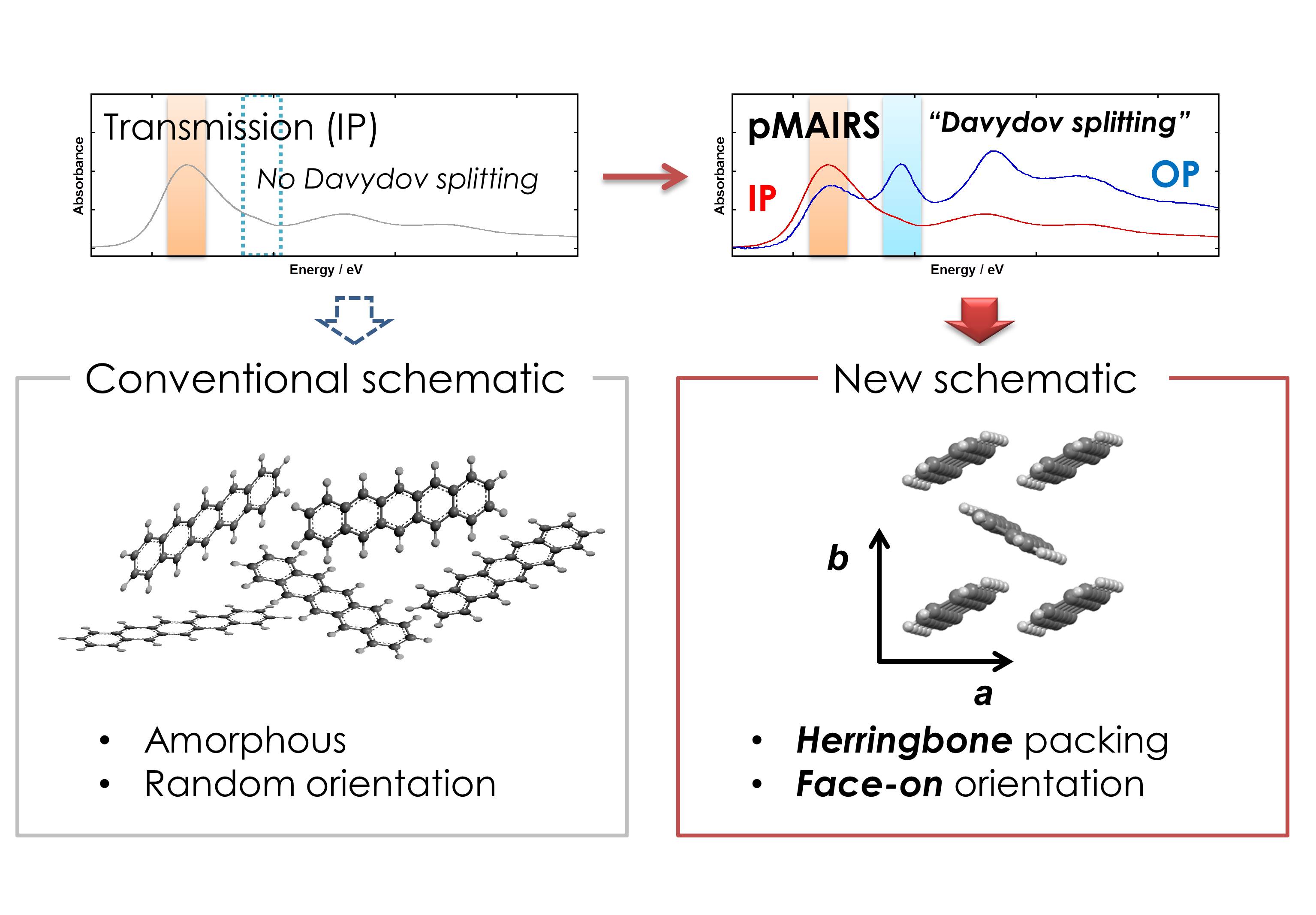
Organic semiconductor materials in optoelectronic thin-film devices form a unique molecular aggregated structure with respect to the molecular orientation and polymorphs. The aggregation structure, optoelectronic property and their relationship have been attracting great interests for a long time in the field of organic electronics. Both control and analysis of the molecular orientation and packing are thus of great importance to understand the relationship. Since organic semiconductor small molecules involving a rigid π-conjugated skeleton typically have a specific molecular orientation to the primary chemical structure, however, selectively control of the molecular arrangement in thin films has been believed impossible. For example, pentacene known as a representative organic semiconductor readily forms a highly ordered structure when deposited on a solid surface, and at the same time the molecular long axis aligns perpendicularly to the substrate surface. The face-on orientation, where the ring plane has a parallel stance, was found limitedly on a surface-active substrate. Preparation of the face-on thin film structure on a practical substrate represented by silicon is challenging.
Our previous studies report that the face-on orientation in organic semiconductor thin films is often observed as a kinetically restricted metastable phase. This straightforwardly gives us a hypothesis that the small cluster favors the face-on orientation. In this sense, the face-on orientation of pentacene should correspond to an intermediate phase between the crystal and amorphous phases.
To confirm this model, a low-temperature thin film of pentacene was prepared and the molecular anisotropic structure was analyzed by using multiple cutting-edge techniques of p-polarized multiple angle incidence resolution spectrometry (pMAIRS) and two-dimensional grazing incidence X-ray diffraction (2D-GIXD). The combined techniques demonstrated that the face-on orientation is definitely realized on a silicon substrate as the dominant component. This concept is believed to be useful for controlling the molecular orientation in general organic semiconductor thin films.
 Institute for Chemical Research, Kyoto University
Institute for Chemical Research, Kyoto University International Joint Usage Research Center
International Joint Usage Research Center